How To Remove Silica From Water?
Finding the best method
There is not a single “one size fits all” answer; I wish it was that simple! There are various ways to remove silica from a water supply, including lime softening, ion exchange, reverse osmosis, ultrafiltration, and electrocoagulation. Most of these methods are not economically feasible in residential applications.
Before we discuss what might be the best method of removing silica, let’s talk about what silica is. It’s important to understand that silica in a water supply is most often naturally occurring rather than man-made. Some applications require water free of silica, such as:
- Car washes
- Cooling towers
- Boilers
- Semi-conductors
- Wastewater
In commercial and industrial applications, the best method to remove silica is most likely determined on a case-by-case basis, taking into consideration the totality of the circumstances, water quality, flow rate, gallons per day, and space requirements. In residential applications, due to cost and space constraints, the choices are a little clearer.
What is Silica?
Also known as silicon dioxide, or SiO2, silica is a crystalline compound that is an extremely common constituent in most rocks, minerals, and sand. Silica is formed by silicon and oxygen with another metal or mineral. Its levels range from 1 part per million to more than 100 parts per million. Typically, silica exists in two forms in the water supply: (1) reactive silica; and (2) colloidal silica. Silica causes etching, scratching, and spotting on glassware and other fixtures.
Removal of silica is usually done as a means of preventing wear and damage to equipment. This is because silica can accumulate on surfaces and appear as hard mineral deposits known as scale. Scale deposits will reduce the efficiency of heating and cooling and interfere with the ability to operate systems within a defined pressure range. Silica scaling can compromise the efficiency, performance, and safety of water using equipment such as boilers, turbines, piping, and even filtration systems.
Silica is most commonly found on Earth in the form of quartz. It is also a significant component of sand. In fact, after oxygen, silicon is the second most abundant element present on our planet. The chemical composition of silica is SiO2.
Silica is frequently confused with hardness. Sometimes when a water softener is installed, it may seem that there still is hardness in the water because of etching or spotting. You can perform a very simple test to determine if it is hardness or silica. Just apply vinegar to the stains, and if they are removed by the vinegar, it is hardness. If the vinegar does not remove it, it is most likely silica.
Let’s look at the most common methods of dealing with and removing silica.
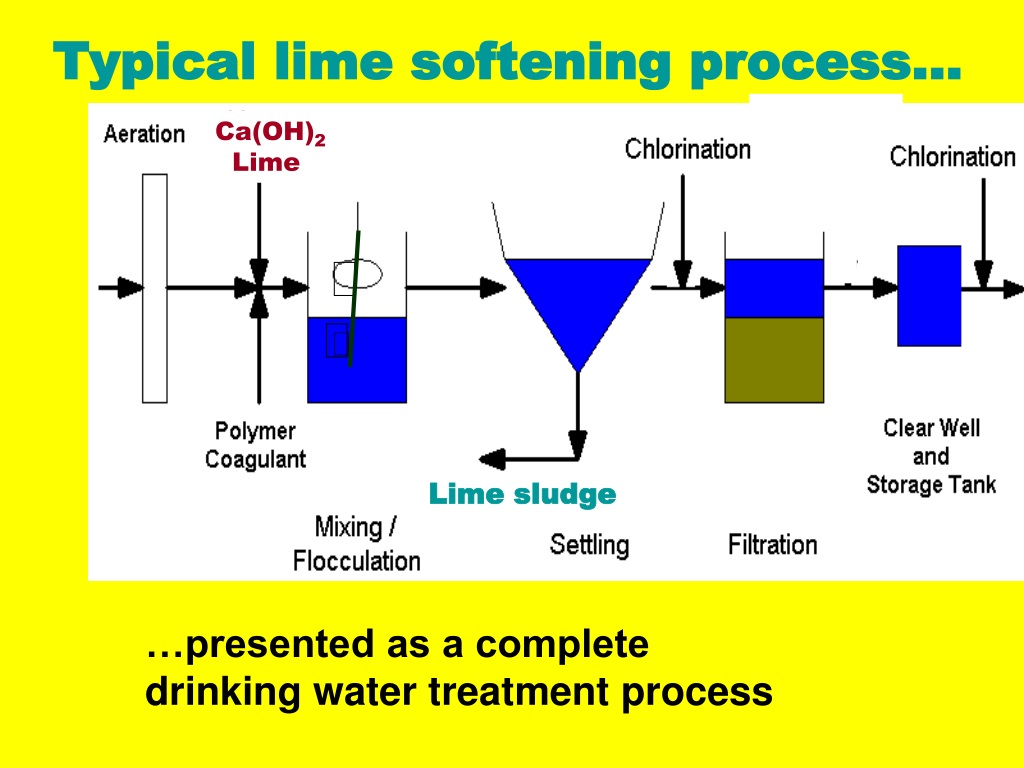
Lime Sofftening
Lime softening requires the addition of calcium hydroxide to the water supply. This causes the calcium carbonate and magnesium carbonate (commonly called “hardness”) to precipitate out. The silica particles are absorbed by the calcium and magnesium hydroxides. The most common applications for lime softening are in large cooling towers, and large boiler feeds.
This method requires a large capital investment and is very costly due to the chemicals needed and the sludge generated, which requires disposal. This is a method that would not be economically practicable in a residential application.
Ion Exchange
Silica molecules have a negative charge, so anion resin is needed to remove them. The biggest problem is that the anion resin needs to be regenerated with caustic soda (NaOH), which is a huge safety concern, especially in a residential application. Because of that, ion exchange is not recommended in residential applications.
Ultrafiltration
Ultrafiltration or UF will only remove colloidal silica, so you must first determine whether your silica is colloidal or reactive silica. Colloidal silica is typically found in surface waters and has created problems for water treatment because of its stability as an un-ionized compound, making it difficult to remove using ion exchange processes.

Reverse Osmosis
If the silica is reactive silica, then Reverse Osmosis is the most practical method to remove it from the water supply. Reverse osmosis will reduce both colloidal and reactive silica, but proper pretreatment is necessary, including oxidation of iron, sulfur, and manganese, removal of chlorine, and chloramine, while hardness must be treated with a water softener or an Anti-Scalant. It is also recommended that the RO system incorporate a permeate flush, which means that the membranes are soaked with RO water, so that scale, including silica, will not foul the membrane surface.

Electrocoagulation
This is an emerging technology that shows a great deal of promise but on an industrial or municipal level. It seems doubtful to me that it would ever be utilized in a residential application.
Summary
Removing silica in a commercial or industrial application will often be different depending upon several variables. However, reverse osmosis with permeate flush and proper pretreatment is the leading technology, as it removes both forms of silica.
If you need more information on how to remove Silica in water, please contact us immediately with the following information:
KHANG NGOC ENVIRONMENT JOINT STOCK COMPANY
Address
Main office:
86 Dong Ho Street, Ward 4, District 8, Ho Chi Minh City, Vietnam
Tel: +8428. 37584648
Mobile: +84983.152.032
Branch office address:
C7/30C14 – Pham Hung Str. – Binh Chanh Dist. – HCM
Tel: +8428.37584648 / +8428. 62755032
Fax: +8428.37583936
Mobile: +84983.152.032
Factory & warehouse:
17 – Pham Hung street – Phuoc Loc ward, Nha Be district – Ho Chi Minh City, Vietnam.
 Business
Business




